Dipl.-Inf. Rüdiger Bock
Alumnus of the Pattern Recognition Lab of the Friedrich-Alexander-Universität Erlangen-Nürnberg
Automated Glaucoma Risk Index
Glaucoma Risk Index: Automated glaucoma detection from color fundus images
In: Medical Image Analysis (2010), 14(3), 471-481 (doi:10.1016/j.media.2009.12.006, BiBTeX)
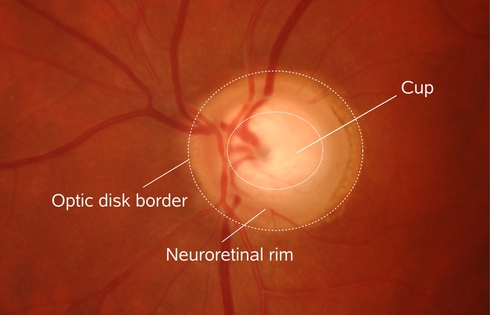 |
Glaucoma is one of the most common causes of blindness with a mean prevalence of 2.4 % for all ages and of 4.7 % for ages above 75 years []. The disease is characterized by the progressive degeneration of optic nerve fibers and astrocytes showing a distinct pathogenetic image of the optic nerve head.
Glaucoma leads to
- structural changes of the optic nerve head (ONH) and the nerve fiber layer and
- a simultaneous functional failure of the visual field.
The structural changes are manifested by a slowly diminishing neuroretinal rim indicating a degeneration of axons and astrocytes of the optic nerve (Figure 1).
This contribution provides a novel Glaucoma Risk Index (GRI) from fundus images by capturing ONH changes caused by glaucoma disease.
Contrary to the established detection techniques, it does not require accurate measurements of geometric ONH structures as it performs a statistical data mining technique on the image patterns themselves.
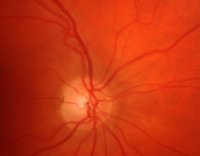
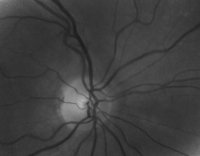

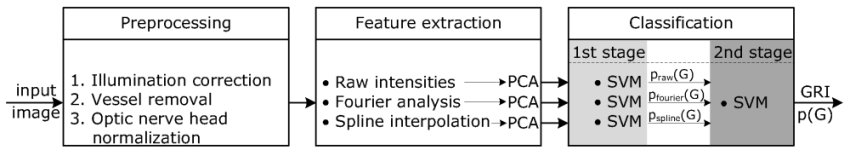
The major procedure illustrated in Figure 2 consists of three steps:
Preprocessing:
The appearance-based techniques preserve the data variation in the low dimensional representation independent from its origin although it might not be related to the classification task.
Variations such as
- illumination inhomogeneities
- ONH location
- vessel structure
are not linked to the glaucoma disease and have to be excluded from the image data beforehand (Figure 3).
Feature extraction:
We propose different types of data representation in order to capture the image information:
- Raw pixel intensities
- Fourier coefficients
- Spline coefficients
These feature types are then compressed separately by PCA to gain a low dimensional image representation for classification.
Classification:
In the last processing step, a probabilistic two-stage classifier scheme using the Support Vector Machine (SVM) combines the different types of features to gain one single glaucoma prediction, the Glaucoma Risk Index (GRI).
 |
Data:
- Erlangen Glaucoma Registry (EGR)
- Gold standard by experienced ophthalmologist
- Kowa NonMyd alpha (optic nerve head centered, 25° FOV)
- Data set
- 239 glaucomatous
- 336 healthy controls
- Evaluation: 5-fold cross validation setup
Comparison to experts and established glaucoma parameters
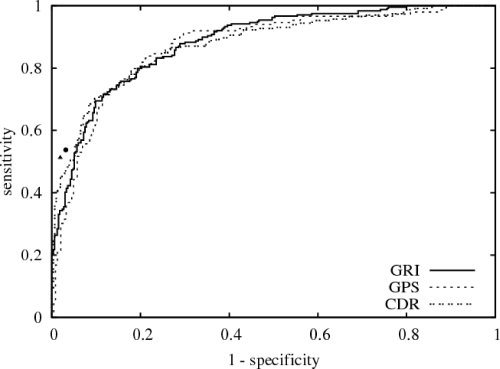
The Reveiver operating characteristic (ROC) curve of GRI (---) with an area of 88 % in comparison:
- Human experts on single fundus images without medical history are very specific while sensitivity reaches a moderate level.
(Spec: 97 %, 98 %; Sens: 54 %, 51 %) - Competitive performance compared to Glaucoma Probability Score (GPS) of Heidelberg Retina Tomograph v3 (HRT) and linear cup-to-disk ratio (CDR) in particular for a specificity around 0.8.
Area under the ROC curve:- GPS: 87 %
- CDR: 88 %
This contribution provides a competitive, reliable and probabilistic glaucoma risk index
from images of the low-cost digital color fundus camera
as its performance is comparable to medical relevant glaucoma parameters.
This proves, data-driven GRI is able to extract relevant glaucoma features. In the
future, it might give a first, low-cost glaucoma indication to route the patients to
more elaborate clinical trials only if necessary.